[最新] homogeneous and heterogeneous mixture diagram 289094-Homogeneous and heterogeneous mixture venn diagram
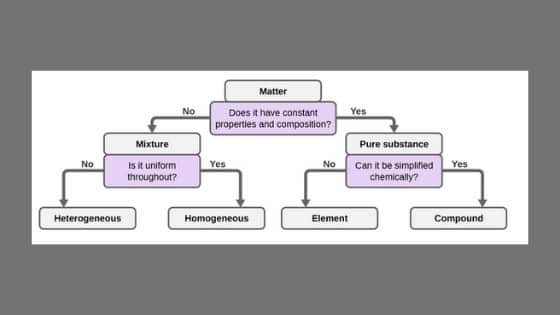
•Explain the difference between homogeneous and heterogeneous mixtures •Know how to represent particle of matter in simple diagrams •Interpret particle diagrams An atom is the smallest part of matter that can exist on it own made up of smaller particles called protons, The difference between homogeneous and heterogeneous mixture are tabulated below Homogeneous Mixture Heterogeneous mixture They have uniform compositions They have nonuniform compositions The components of homogeneous mixtures are not physically distinct A heterogeneous mixture has physically distinct componentsHeterogeneous mixtures have nonuniform composition What type of mixtures are these?
How To Distinguish Pure Substances And Mixtures Dummies
Homogeneous and heterogeneous mixture venn diagram
Homogeneous and heterogeneous mixture venn diagram-About heterogeneous mixture homogeneous mixture worksheet The heterogeneous mixture – homogeneous mixture worksheet with answer key is below The worksheet gives common examples of mixtures, in addition to some pure, unmixed substances Learn how to classify these examples of mixtures belowThis chemistry video tutorial explains the difference between homogeneous and heterogeneous mixtures within the subtopic of the classification of matter It




Classification Of Matter Chemistry Grade 10 Caps Openstax Cnx
What Is a Homogeneous Mixture? Examples of homogeneous mixtures include pure water, corn oil, blood plasma, steel alloys, and air ADVERTISEMENT Before we tackle the individual examples of homogeneous mixtures, let's take a closer look at the definition of homogeneous mixtures and contrast them with heterogeneous mixturesHomogeneous and heterogeneous azeotropes If the constituents of a mixture are completely miscible in all proportions with each other, the type of azeotrope is called a homogeneous azeotrope For example, any amount of ethanol can be mixed with any amount of water to form a homogeneous solution
Heterogeneous refers to a structure with dissimilar components or elements, appearing irregular or variegated For example, a dermoid cyst has heterogeneous attenuation on CT It is the antonym for homogeneous, meaning a structure with similar components Heterogenous refers to a structure having a foreign originAs students become familiar with these categories, they will be able to use a tree diagram to map the examples LEARNING OBJECTIVE Students will differentiate pure substances (chemicals) and mixtures Students will differentiate homogeneous mixtures (solutions) and heterogeneous mixtures Students will differentiate elemental substances and Homogeneous → sodawater, wood, air, alcohol and water mixture, sugar and water mixture, salt and water mixture, cough syrup Heterogeneous → soil, petrol and water mixture, chalk and water mixture, sand and water mixture, pumice stone, smoke, gun powder The correct answer was given riddhima95 answer
Homogeneous blends just have one stage gas, fluid or strong Different homogenous blends are air, water and vodka Heterogeneous Mixtures Heterogeneous blends are comprised of noticeably extraordinary substances or stages A suspension is a kind of heterogeneous blend with huge particles, obvious Heterogeneous mixtures of intermediatesized particles that do not settle out are known as colloids The most abundant substance in a colloid isCheerios Trail mix Trail mix is HETEROGENEOUS Homogeneous Mixtures Heterogeneous Mixtures centrifugation coagulation distillation evaporation filtration hand picking magnetic separation sieving winnowing sedimentation



Mixtures Images Stock Photos Vectors Shutterstock




Using A Venn Diagram Write The Characteristics Of Homogeneous And Heterogeneous Mixture Brainly Ph
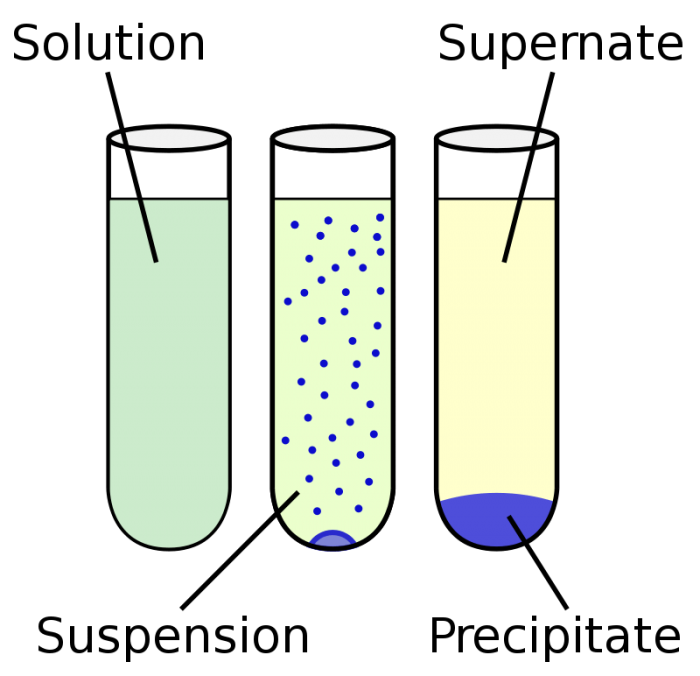
Examples of Homogeneous and Heterogeneous Mixtures Examples of heterogeneous mixtures would be ice cubes (before they melt) in soda, cereal in milk, various toppings on a pizza, toppings in frozen yogurt, a box of assorted nutsEven a mixture of oil and water is heterogeneous because the density of water and oil is different, which prevents uniform distribution in the mixtureA mixture having nonuniform composition A mixture having the same composition and uniform appearance throughout A colloid can either be heterogeneous or homogeneous In a colloid the particles are spread throughout These particles are small, well mixed, an don't settle to the bottom Mixtures SuspensionHomogeneous mixtures exist in one phase of matter at a time You will not see liquid water and solid water together in a homogeneous mixture That means your glass of ice water, with ice cubes floating in it, is a heterogeneous mixture of homogeneous mixtures Homogeneous mixtures cannot be expressed as chemical formulas




Heterogeneous Homogeneous Mixtures




Determine Whether Each Molecular Diagram R Clutch Prep
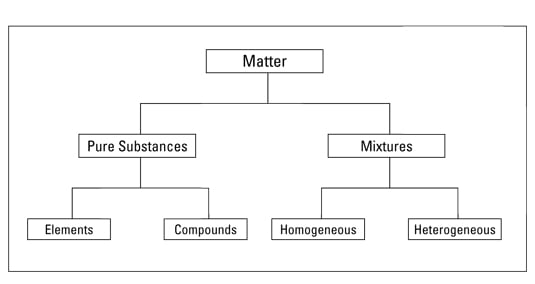
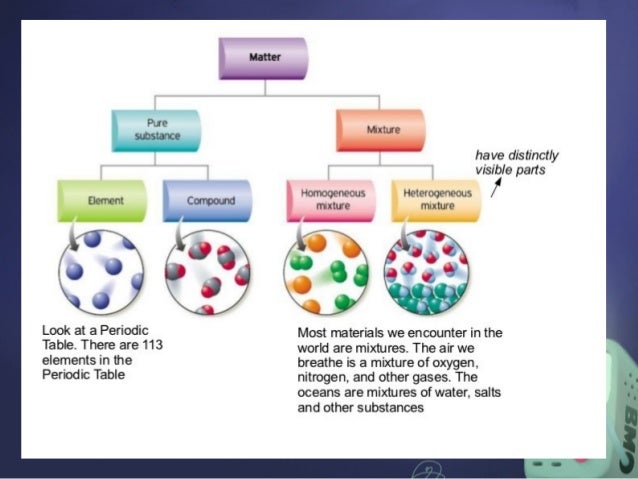
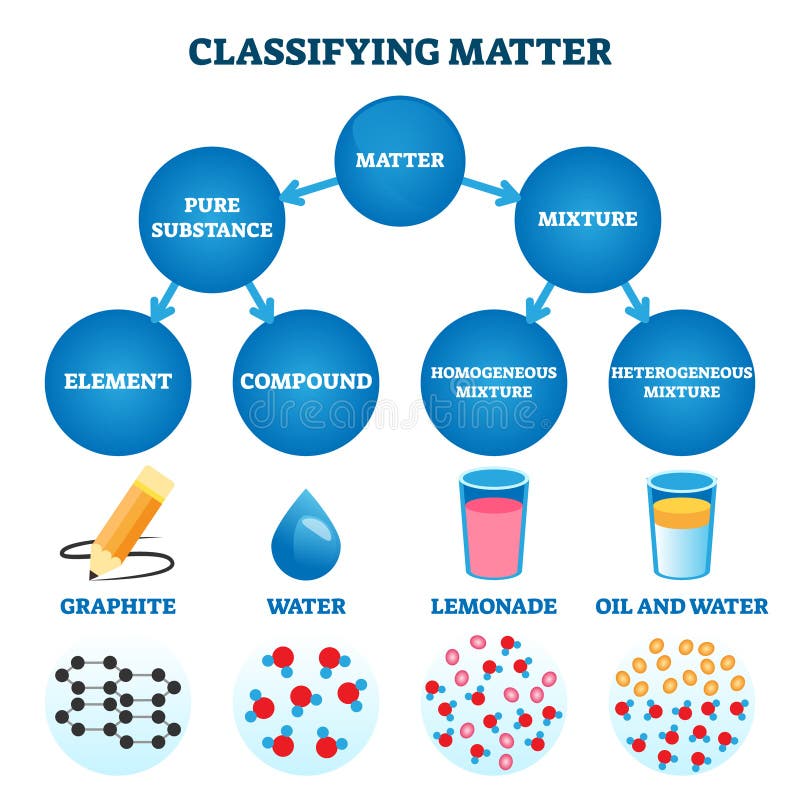
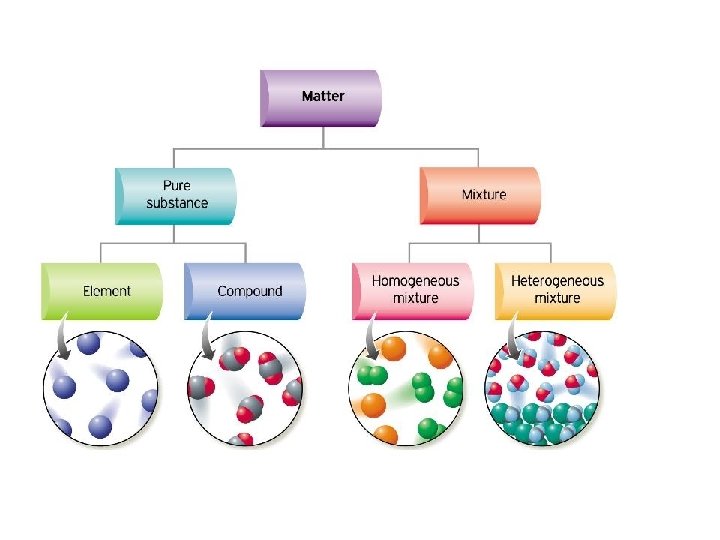
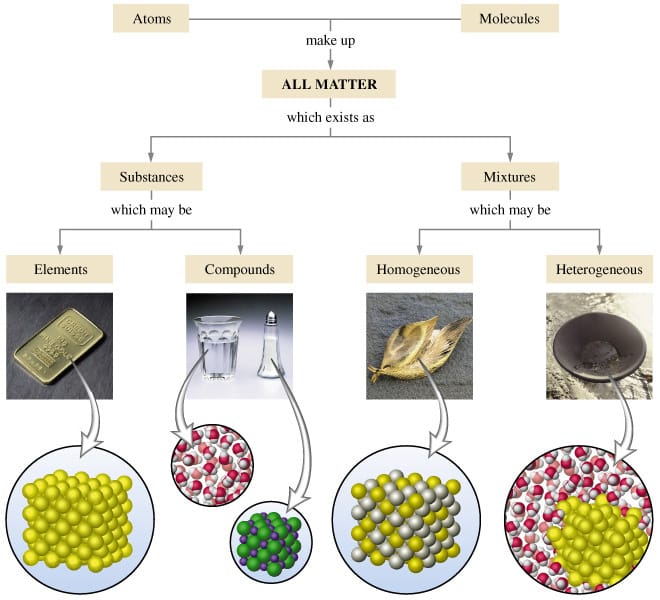
Heterogeneous mixture is a mixture with a nonuniform composition When you mix two components that remain separate from each other, that mixture is called a Heterogeneous mixture Concrete is an example of a Heterogenous mixture A mixture of Cement and Water A mixture of cold drinks and ice cube is also an example of a Heterogeneous mixtureThe major advantages/disadvantages of heterogeneous vs homogeneous catalysts are summarized in Table 1 As a general picture, the main difference is the fact that in the case of homogeneous catalysts, every single catalytic entity can act as a single active site This makes homogeneous catalysts intrinsically more active and selectiveA diagram representing at the microscopic level the differences between homogeneous mixtures, heterogeneous mixtures, compounds, and elements Solution edit A solution is a special type of homogeneous mixture where the ratio of solute to solvent remains the same throughout the solution and the particles are not visible with the naked eye




Pure Substances And Mixtures Ppt Download



Chapter 10 1 Mixtures Solubility Acid Base Solutions
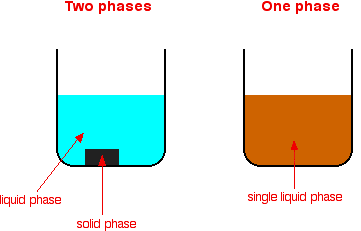
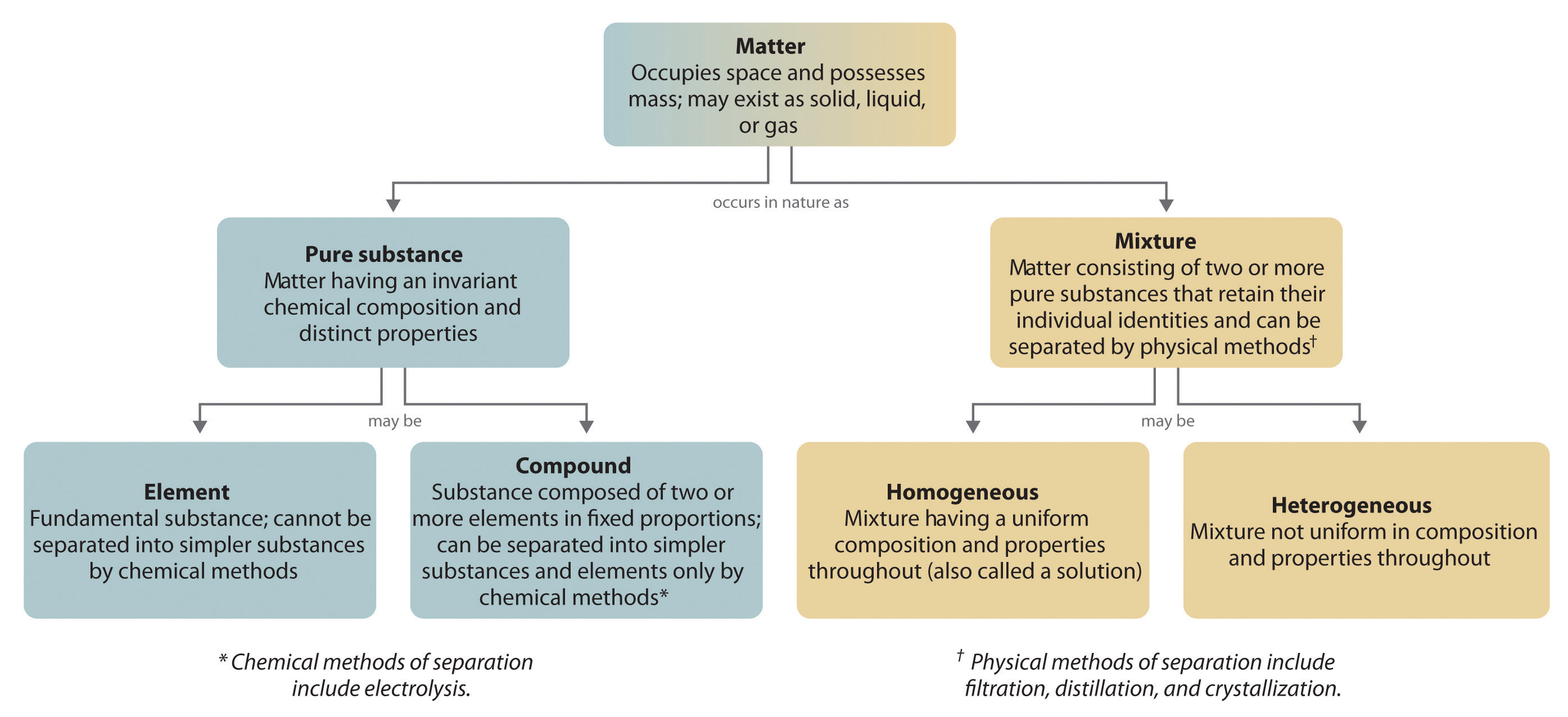
A homogeneous mixture has the same uniform appearance and composition throughout Many homogeneous mixtures are commonly referred to as solutions A heterogeneous mixture consists of visibly different substances or phases The three phases or states of matter are gas, liquid, and solidBy definition, a pure substance or a homogeneous mixture consists of a single phase A heterogeneous mixture consists of two or more phases When oil and water are combined, they do not mix evenly, but instead form two separate layers Each of the layers isA diagram representing at the microscopic level the differences between homogeneous mixtures, heterogeneous mixtures, compounds, and elements Solution A solution is a special type of homogeneous mixture where the ratio of solute to solvent remains the same throughout the solution and the particles are not visible with the naked eye, even if



3




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii
This video is in simple language about Difference between homogenous and heterogeneous mixturesClass 9Chapter 2Is Matter Around Us Pure Homogeneous and Heterogeneous Mixtures the differences between Heterogeneous and Homogeneous mixtures You can edit this template and create your own diagram Creately diagrams can be exported and added to Word, PPT (powerpoint), Excel, Visio or any other document #Venn #VennDiagram #VennDiagramExamples #VennTemplates The homogeneous mixture is the combination of two or more pure substance in such a uniform manner that each of the substance is indistinguishable from the other substance, whereas the pure substances in the heterogeneous mixture are not uniformly distributed, and it results in the formation of nonuniform composition




Classification Of Matter Chemistry Grade 10 Caps Openstax Cnx
.png?revision=1)



1 3 Classification Of Matter Chemistry Libretexts
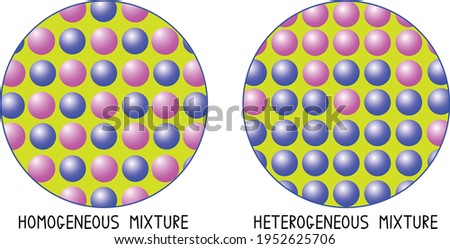
So, we could label the diagram on the right as a homogeneous mixture because the particles are all evenly distributed If heterogeneous is the opposite, that must mean that in a heterogeneous mixture, we have unevenly distributed particles A mixture consists of sugar in water is a homogeneous mixture because we can't see particles of sugar in the water, as they are dissolved thoroughly A mixture consisting of oil in water is an example of the heterogeneous mixture as the oil cannot be mixed in the water and we can easily see them I hope you like my post about "DifferenceUse 42 colorful cards to sort examples of mixtures and solutions, sort homogeneous or heterogeneous mixtures, identify the solvent and solute in solutions and choose separation methods for 16 different mixtures Includes printable version for inclass use and a google slides version that can be sha Subjects




Mixture Wikiwand




How To Distinguish Pure Substances And Mixtures Dummies
In a homogeneous mixture, the particles are dispersed evenly throughout In other words, a homogeneous mixture is uniform A sample from one part of a homogeneous mixture is the same as any other sample of the mixture A homogeneous mixture looks uniform even under a microscopeINSERT THE TITLE OF YOUR PRESENTATION HERE Heterogeneous and Homogeneous Mixtures Examples ALLPPTcom _ Free PowerPoint Templates, Diagrams and Charts I I OBJECTIVES A Content Standards The learners demonstrate understanding of different types of mixtures and their characteristics BMixtures are made up of two or more substances which are the constituents of mixture The constituents of mixture can be in any ratio Mixtures can be divided into 1 Homogeneous mixtures 2 Heterogeneous mixtures Homogeneous mixtures have uniform composition For instance, sugar in water, water in alcohol etc




Is Salty Water Homogeneous Or Heterogeneous




Chapter 6 Venn Diagram By Charlie Lemkuil
A type of homogeneous mixture where the particles of one substance are dispersed or suspended in another trail mix an example of a heterogeneous mixture steel or brass example of an alloy magnetic attraction, evaporation, filtration, distillation methods to separate mixtures not combined in a set ratio diagrams flashcards mobileClassify the following as homogeneous or heterogeneous Learn with flashcards, games, and more — for freeShow all show all steps Step 1 of 3 A mixture that has a uniform composition throughout the sample is called a homogenous mixture Comment ( 0) Chapter 2, Problem 59P is solved




Homogeneous And Heterogeneous Mixtures Youtube




Shutterstock Puzzlepix
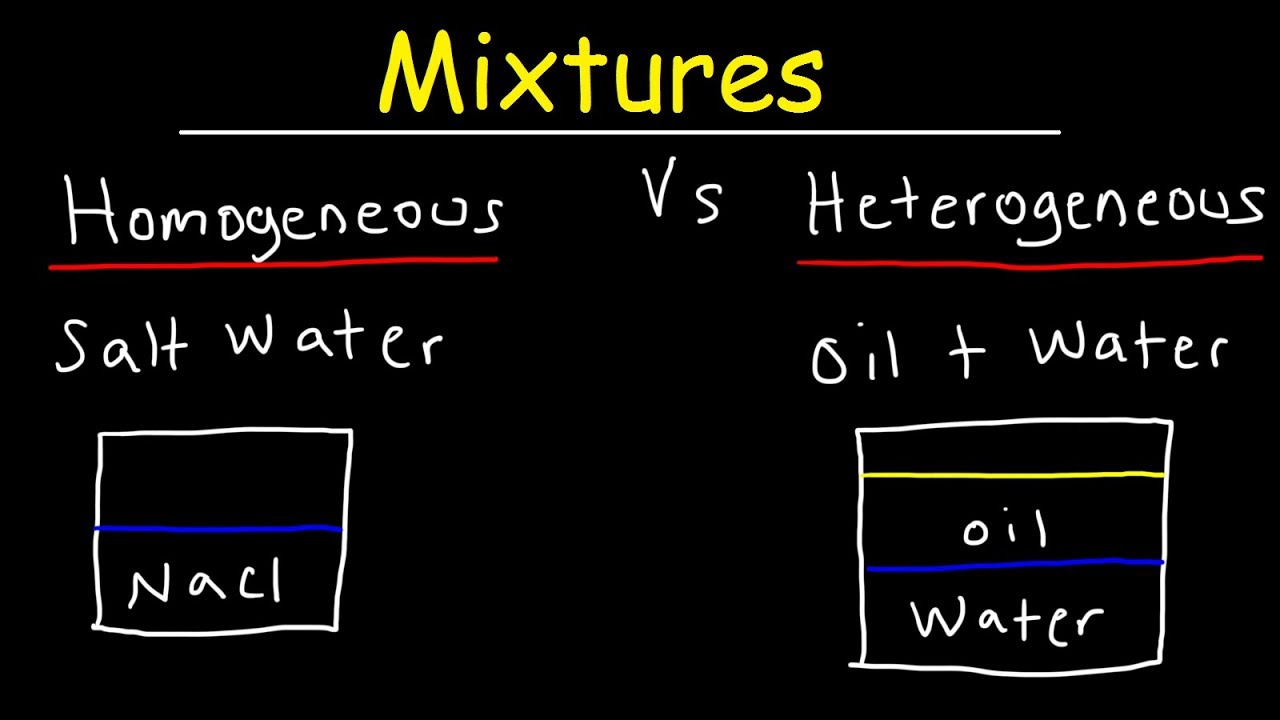
Homogeneous mixture Heterogeneous mixture 1) These are called as solutions These are called as suspensions/colloids 2) Substances are Uniformly distributed These substances are Unevenly distributed 3) These are not visible to the naked eye, but visible through the microscopeThis mixture was deeply studied in literature since it is the typical case of aqueousorganic mixture with liquid phase demixing and that shows an heterogeneous azeotrope (x water = 0747 mol/mol) Given the feed composition and the distillation specifications discussed in the previous chapter, the corresponding equilibrium phase diagram is A homogeneous mixture is a mixture in which the components that make up the mixture are uniformly distributed throughout the mixture The composition of the mixture is the same throughout There is only one phase of matter observed in a homogeneous mixture at a time So, you wouldn't observe both a liquid and a gas or a liquid and a solid in a homogeneous mixture




Compare And Contrast Homogeneous And Heterogeneous Mixtures Using The Venn Diagram Below In Filling Brainly Ph



Homogeneous And Heterogeneous Mixtures Examples Classification Of Matter Chemistry Youtube
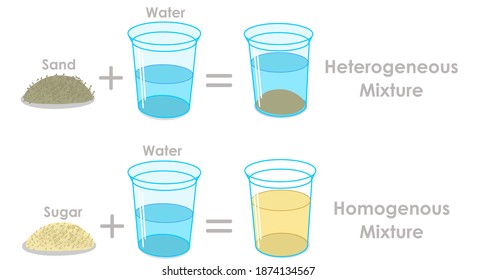
Mixtures A mixture is made from different substances that are not chemically joined For example, powdered iron and powdered sulfur mixed together makes a mixture ofSummary A mixture is a physical blend of two or more components, each of which retains its own identity and properties in the mixture A homogeneous mixture is a mixture in which the composition is uniform throughout the mixture All solutions would be considered homogeneous A heterogeneous mixture is a mixture in which the composition is not uniform throughout the Unlike homogeneous mixtures, heterogeneous mixtures do not have the same composition throughout Oil and water is a fine example of a heterogeneous mixture When oil is mixed with water, the separation of two layers is visible to the eye The following picture shows this phenomenon clearly



Homogeneous Mixture Stock Illustrations 7 Homogeneous Mixture Stock Illustrations Vectors Clipart Dreamstime




Dem Model Configuration A Homogeneous Mixtures B Heterogeneous Mixtures Download Scientific Diagram
4 $395 $295 PDF This 3 page worksheet covers the topics of elements, compounds and mixtures, homogeneous vs heterogeneous mixtures, extensive vs intensive properties, physical and chemical properties, physical and chemical changes, and states of matter Information is presented in a logical progression and has eA homogeneous equilibrium is one in which all of the reactants and products are present in a single solution (by definition, a homogeneous mixture ) Reactions between solutes in liquid solutions belong to one type of homogeneous equilibria The chemical species involved can be molecules, ions, or a mixture of both For the homogenous reaction42 Ternary Mixture Ethanol was then added to the previous components with the consequent introduction of the corresponding waterethanol homogeneous azeotrope in the phase equilibrium diagram A new stable node is present in the phase diagram (ie the homogeneous azeotrope) while pure ethanol is a saddle point




What Is Matter Ck 12 Foundation



Matter Mixtures Mixtures Many Substances In Nature Are
A homogeneous phase is a single phase of matter where everything is in solution Filtered Apple juice that is yellow but transparent is a homogeneous phase Orange juice with pulp floating in it is heterogeneous The pulp is a different phase (solHeterogeneous And Homogeneous Venn Diagram Heterogeneous And Homogeneous Venn Diagram Displaying top 8 worksheets found for this concept Some of the worksheets for this concept are Mixtures and solutions work 3rd grade, Grade 7 science unit 3 mixtures and solutions, Pure mixture practice quiz name key pure or mixture, Venn diagram on solutions andHeterogeneous mixture diagram diagram of heterogeneous mixture heterogeneous mixture particle diagram Latest updated pages Happy Anniversary To You Both Funny Slugterra Megamorph Ramstone Slug Difference Between Heterogeneous & Homogeneous Mixtures pic source pic source




Homogeneous And Heterogeneous Mixed Populations An Example Of A Download Scientific Diagram



What Is A Gaseous Example Of A Heterogeneous Mixture Quora
A chemical mixture combines two substances that maintain their own properties when combined Heterogeneous mixtures are made up of a nonuniform composition, while homogeneous mixtures are made up of a uniform compositionFor example, water and sand is a heterogeneous mixture — you can easily separate the sand from the water



Sci8u1l2




10 Examples Of Mixtures




Mixtures And Solutions Essential Ideas In Chemistry
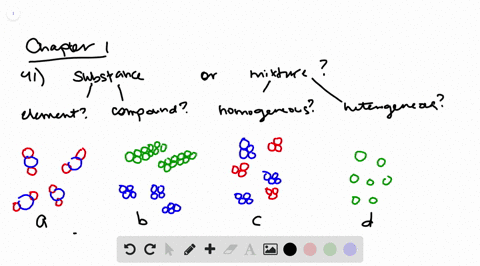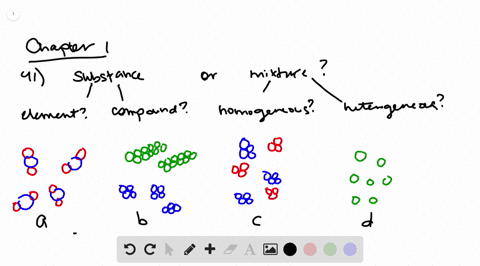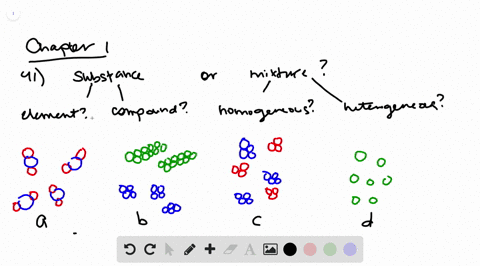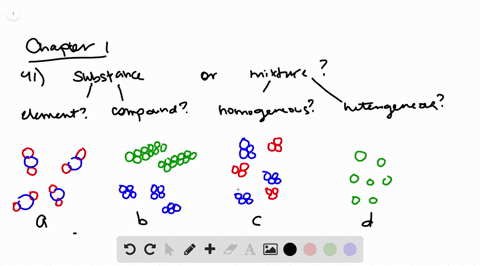



Solved Determine Whether Each Molecular Diagram Represents A Pure Substance Or A Mixture If It Represents A Pure Subst




Label Each Of The Following Four Diagrams In Terms Of Chegg Com



Solution Phase Diagram Shefalitayal



Classifying Matter Schoolworkhelper




Homogeneous Mixture And Heterogeneous Mixture Is Matter Around Us Pure Chemistry Class 9 Youtube




Types Of Matter Flashcards Quizlet



Types Of Catalysis
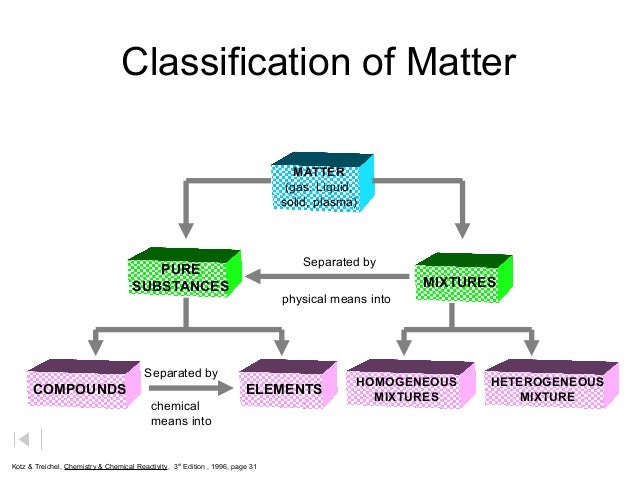



Ch 2 Classification Of Matter Ppt



Pure Substances Mixtures And Solutions Ppt Video Online Download
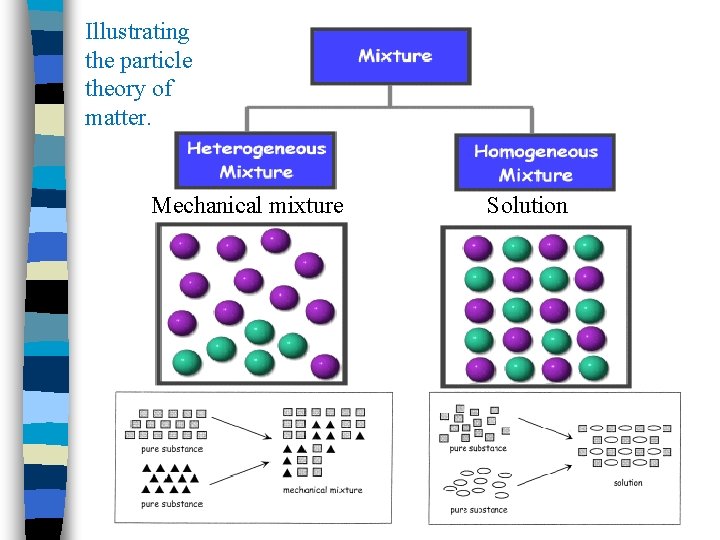



Grade 7 Science Unit 3 Mixtures Solutions Chapter




Homogeneous Vs Heterogeneous Mixtures A Comparison Expii




Homogeneous Mixture Examples In Daily Life




Difference Between Homogeneous And Heterogeneous Material Youtube
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures
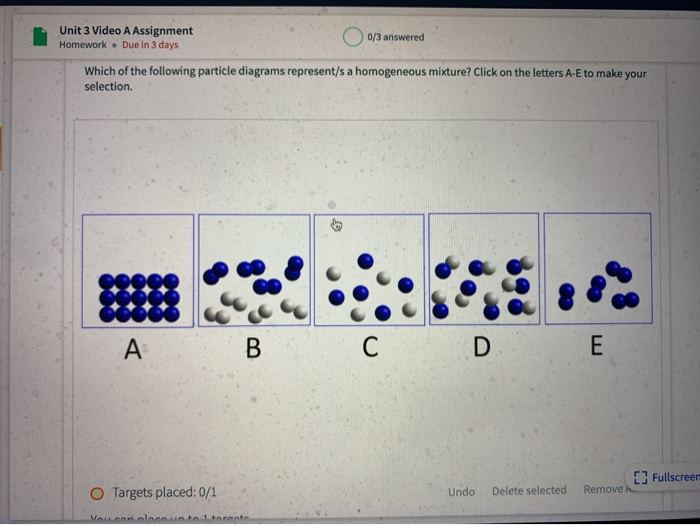



Unit 3 Video A Assignment Homework Due In 3 Days Chegg Com




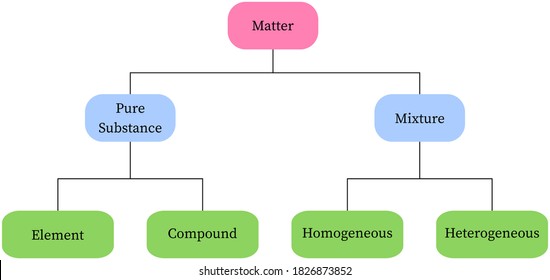
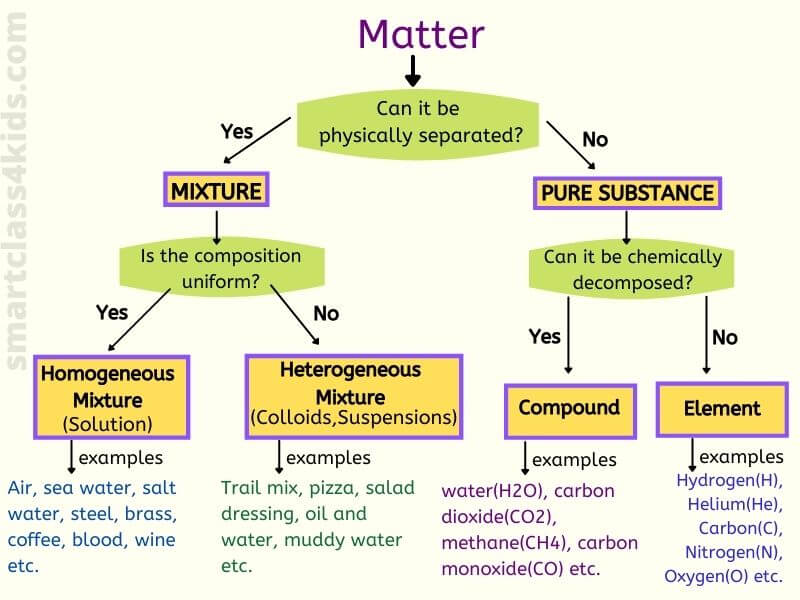
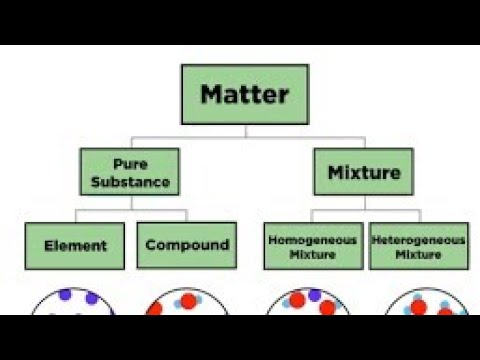
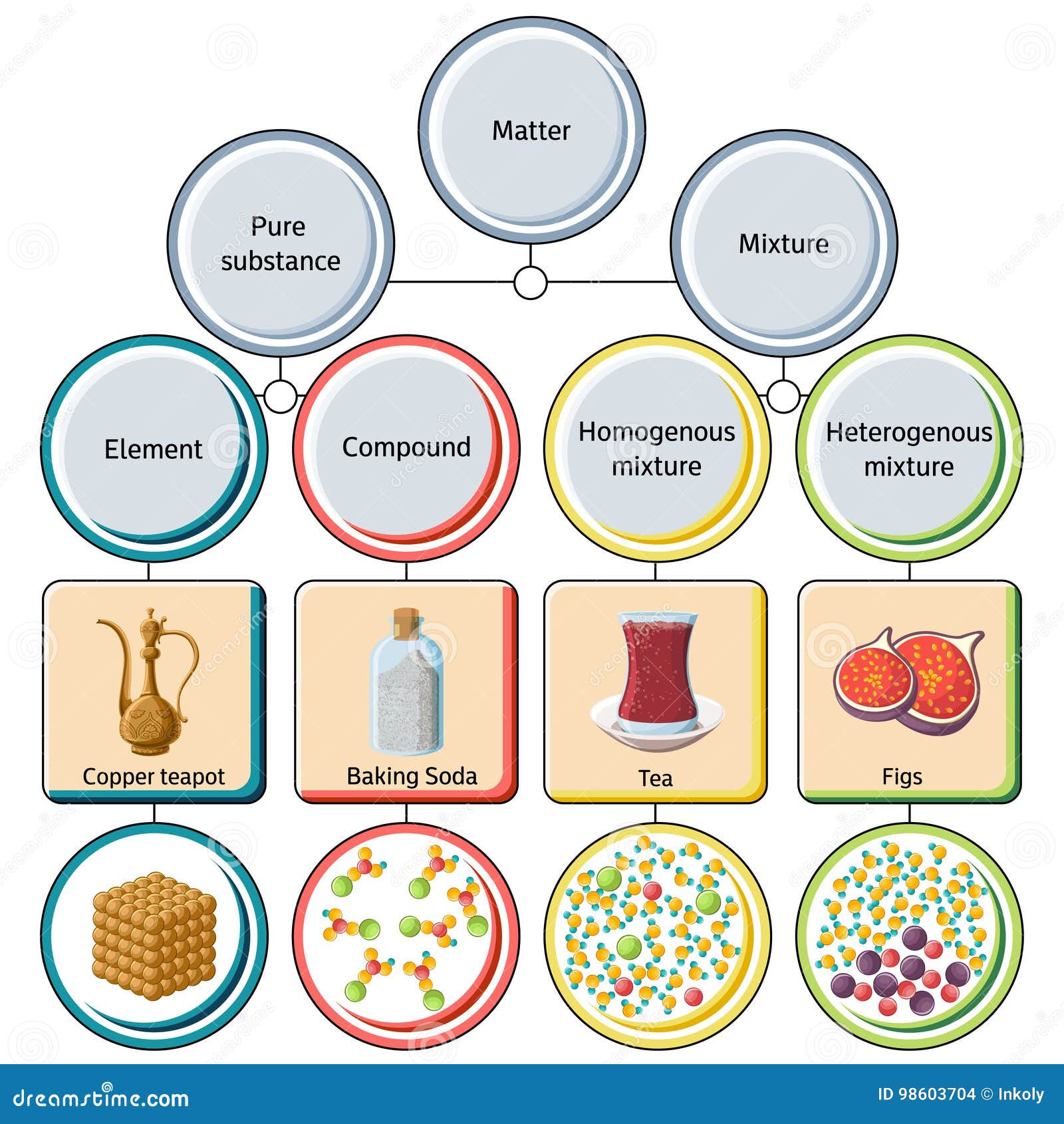
Matter Pure Substance Elementcompound Mixture Homogeneous Solution Colloid Heterogeneous Suspension Solutions Cell Processes Forms Of Matter



Pure Substance High Res Stock Images Shutterstock



What Is Mixture Homogeneous Mixture Heterogeneous Mixture With Examples




Pure Substances Elements Compounds Homogenous Heterogenous Mixture Examples And Problems Youtube



Pure Substance And Mixture Sepration Of Mixtures Mixture Substance Particle Sedimentation Youtube



Homogeneous Mixture Stock Illustrations 7 Homogeneous Mixture Stock Illustrations Vectors Clipart Dreamstime




Difference Between Homogeneous Mixture And Heterogeneous Mixture



A Description Of Matter



Homogeneous Mixture And Heterogeneous Mixture Ncert Books



Self Check Quizzes




Mrs Remis Science Blog 8th Grade Mixtures 13



Shutterstock Puzzlepix




Compare And Contrast Homogeneous And Heterogeneous Mixture Using The Venn Diagram Below Brainly Ph




Given The Particle Diagram Which Type Of Matter Is Represented By The Particle Diagram 1 Brainly Com




Homogeneous Mixtures Vs Heterogeneous Mixtures By Brianna Dearmon



Heterogeneous Mixtures



5 Examples Of Homogeneous Mixture For Chemistry Class Science Trends




Dem Model Configuration A Homogeneous Mixtures B Heterogeneous Mixtures Download Scientific Diagram



1




Let S Talk Chemistry All About Mixtures



Homogeneous And Heterogeneous Mixture Difference Between Homogeneous And Heter Video Dailymotion




Advantages And Disadvantages Of Homogeneous And Heterogeneous Catalysts Download Scientific Diagram




Venn Diagram Create A Venn Diagram For Solution Colloid And Suspension You Will Need To Include Particle Size Type Of Mixture Homogeneous Or Heterogeneous Ppt Download



Homogeneous And Heterogeneous Mixture Nine Science



3
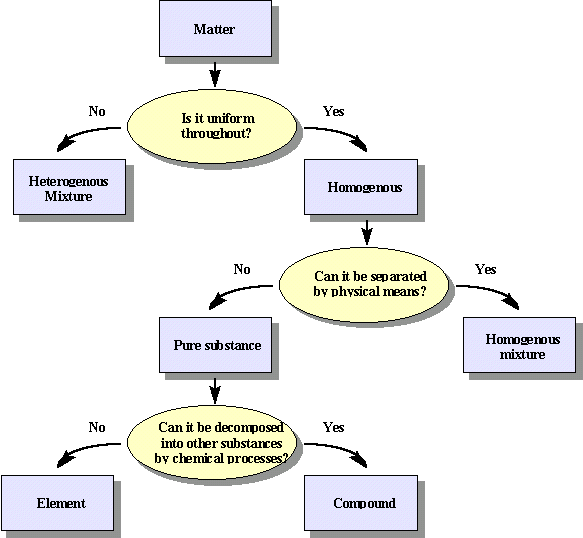



Classification Of Matter Chemistry Libretexts




Ppt Objective I Will Distinguish Between Homogeneous And Heterogeneous Mixtures Powerpoint Presentation Id
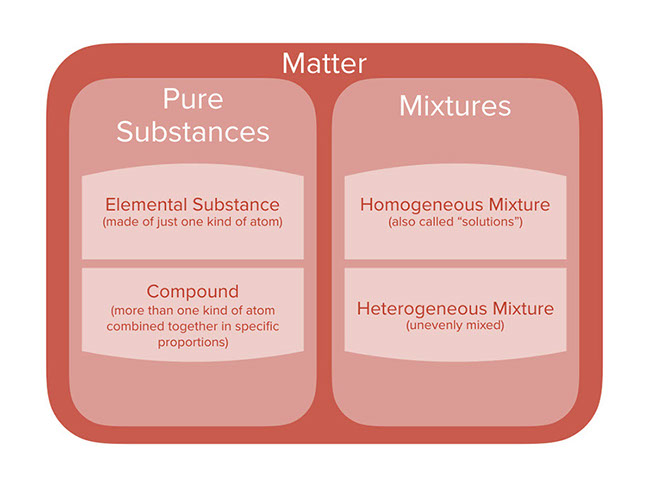



Lesson Categories Of Chemicals And Mixtures




Colloids Chemistry Atoms First 2e




Compound Vs Mixture Difference And Comparison Diffen
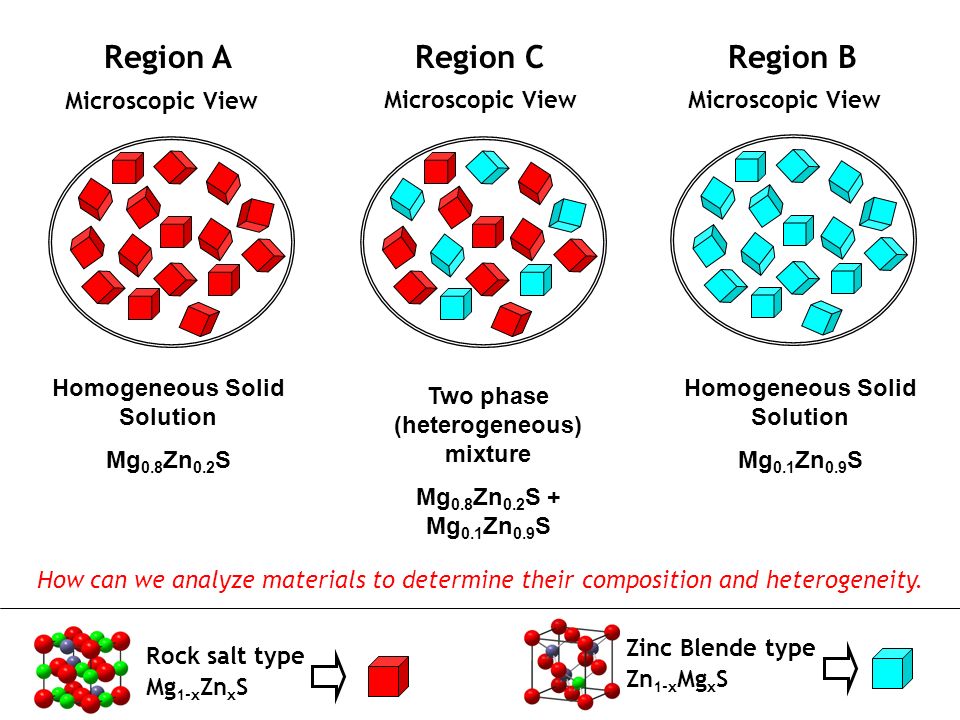



Solid State Synthesis Phase Diagrams And Solid Solutions Chemistry 123 Spring 08 Dr Woodward Ppt Download
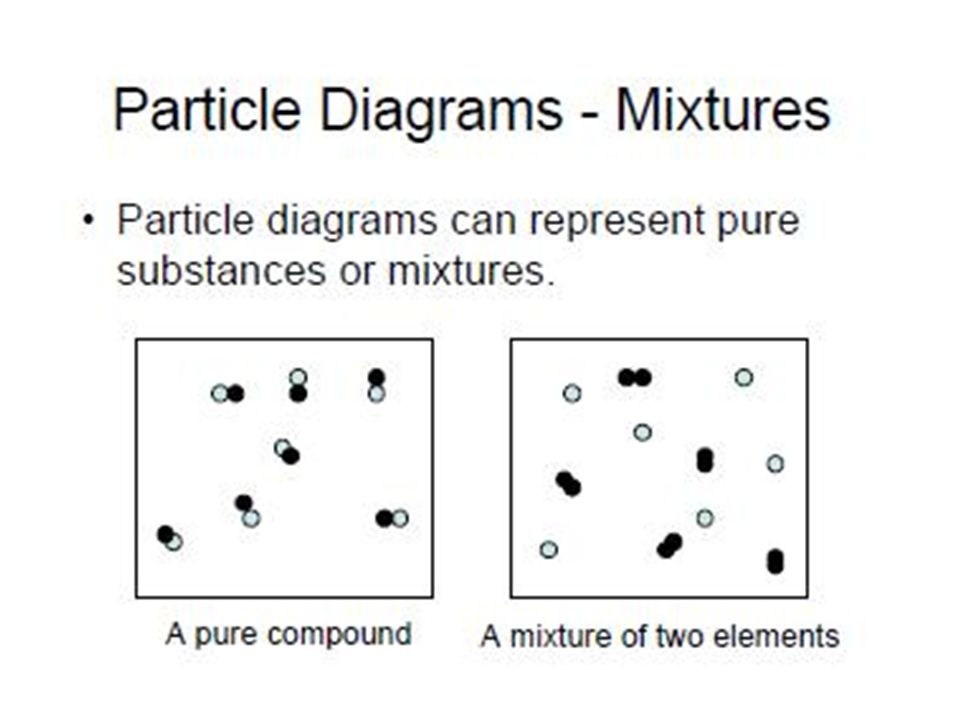



Unit 1 Introduction To Matter 1 2 What Is Particle Diagram Aim How Do You Create A Particle Diagram And How Do You Use It To Distinguish Between A Pure Ppt Download




Pure Substances And Mixtures Review Diagram Quizlet




42 Determine Whether Each Molecular Diagram Chegg Com
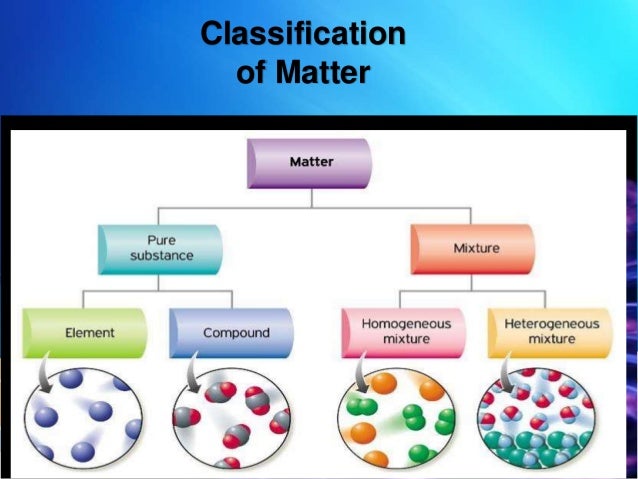



Types Of Mixture




Heterogeneous Mixture High Res Stock Images Shutterstock
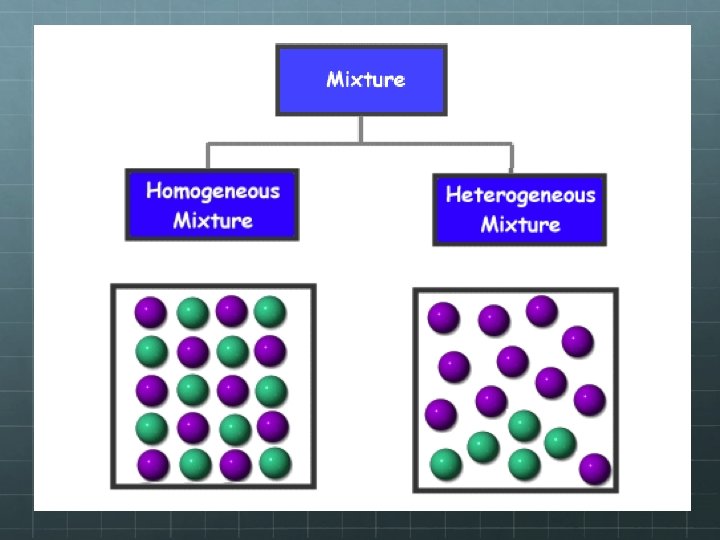



Pure Substances And Mixtures Unit 2 Matter Can




Homogenous Definition And Examples Biology Online Dictionary
/TC_606106-heterogeneous-and-homogeneous-mixtures1-5ac4f1a9642dca0036847e52.png)



Heterogeneous Vs Homogeneous Mixtures
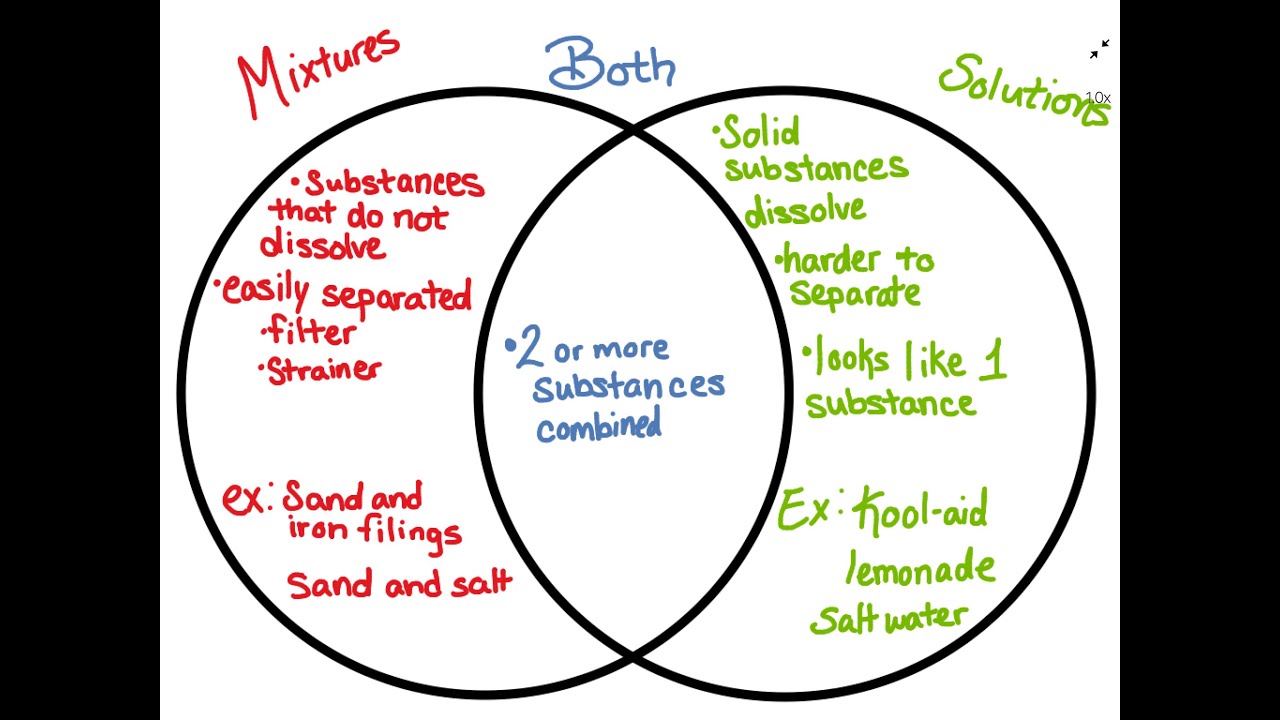



Mixtures And Solutions Venn Diagram Youtube
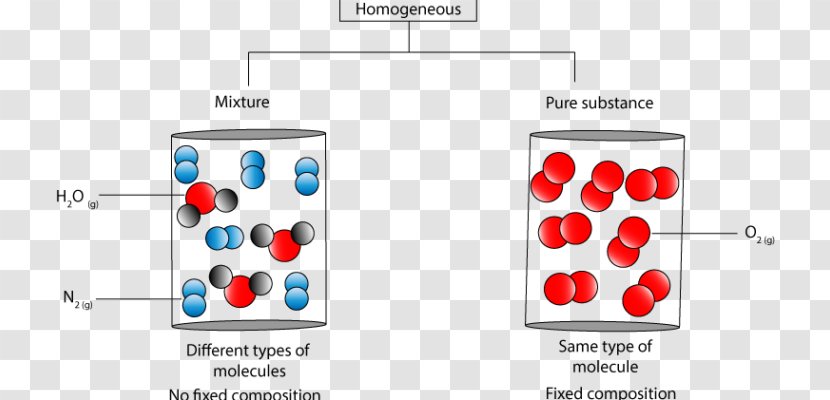



Homogeneous And Heterogeneous Mixtures Chemical Substance Molecule Matter Diagram Red Transparent Png
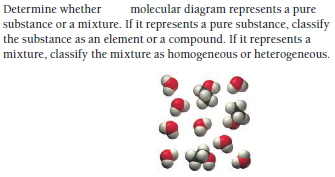



Answered Molecular Diagram Represents A Pure Bartleby




Pure Substances And Mixtures Venn Diagram Examples Venn Diagram Worksheet Venn Diagram Template



Mixtures Ppt Download




Mixture Substances Images Stock Photos Vectors Shutterstock



Matter Practice Quiz Answers
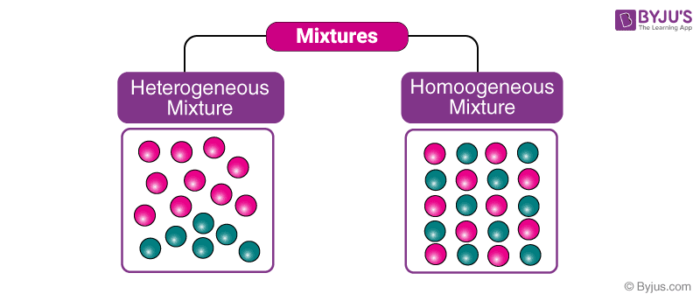



Heterogeneous And Homogeneous Mixture Differences Videos Examples




What Is A Pure Substance Definition Examples Compounds Mixtures Diagram




Classification Of Matter Chemistrygod



Q Tbn And9gctibwptyh6bxmyqlpvsgocys4d1khiseihxu0tbxuqtzfsrfcm8 Usqp Cau
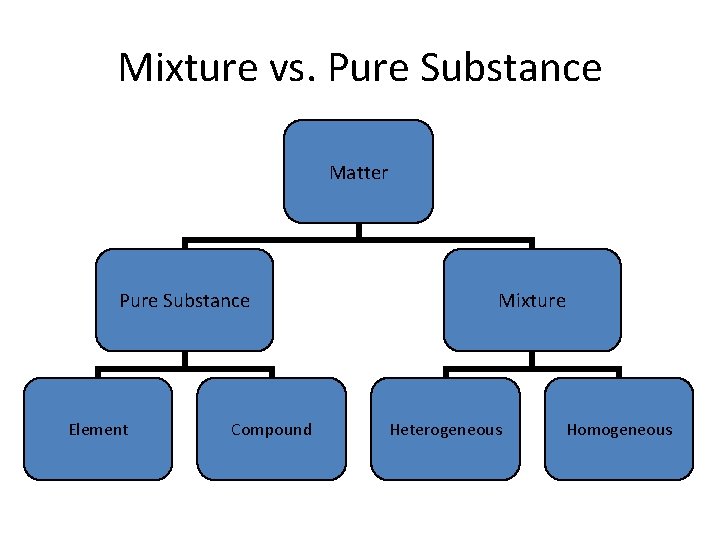



Classifying Matter 6 Mixture Vs Pure Substance Matter
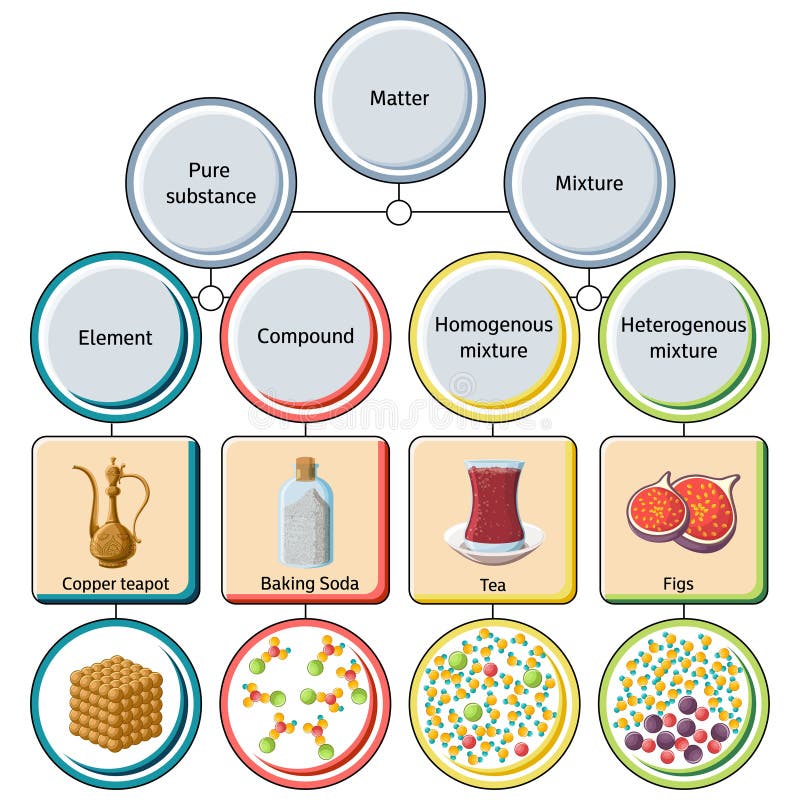



Homogeneous Mixture Stock Illustrations 7 Homogeneous Mixture Stock Illustrations Vectors Clipart Dreamstime




Homogeneous And Heterogeneous Mixtures Editable Venn Diagram Template On Creately




U2cc Particle Diagrams Youtube
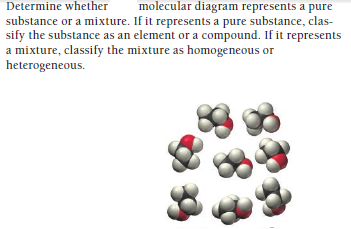



Answered Determine Whether Molecular Diagram Bartleby




The Homogeneous And Heterogeneous Mixture Diagram Quizlet
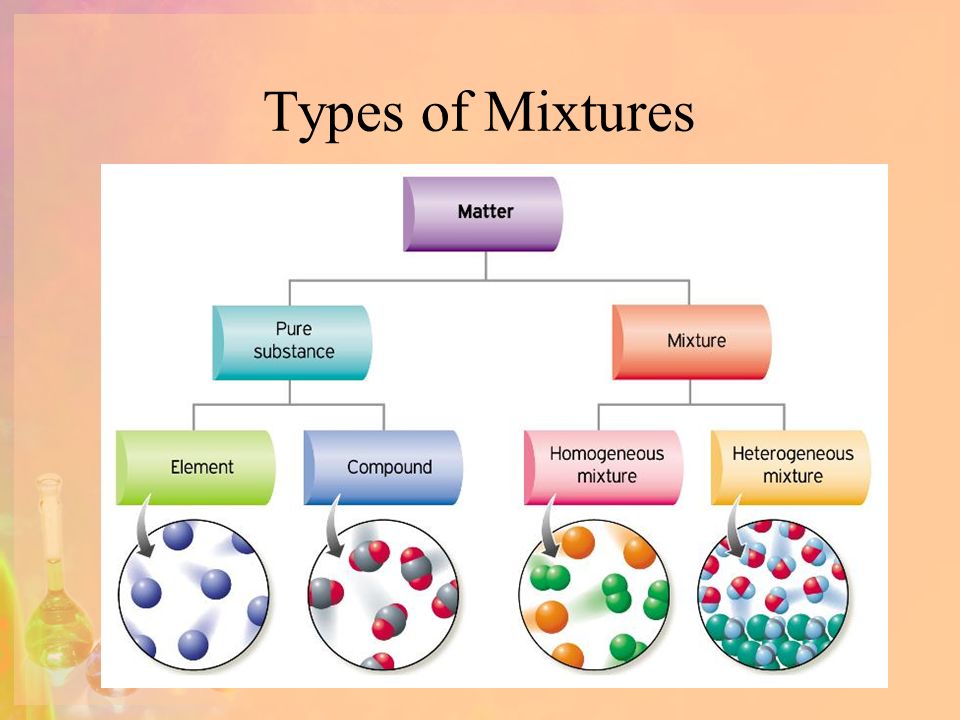



Solutions Types Of Mixtures Objectives 1 Distinguish Between Hetergeneous And Homogeneous Mixtures 2 List Different Solute Solvent Combinations 3 Compare Ppt Download




Chapter 1 Br Section A Br Some Basic Definitions



Is Wood Homogeneous Or Heterogeneous Quora




Homogeneous And Heterogeneous Mixtures Heterogeneous Mixture Venn Diagram Examples Venn Diagram Template




Quality Of The Experimental Varnishes Obtained Green Clear And Download Scientific Diagram




Reasons Provided By Students To Justify Their Classification Of The Download Scientific Diagram
コメント
コメントを投稿